Overview
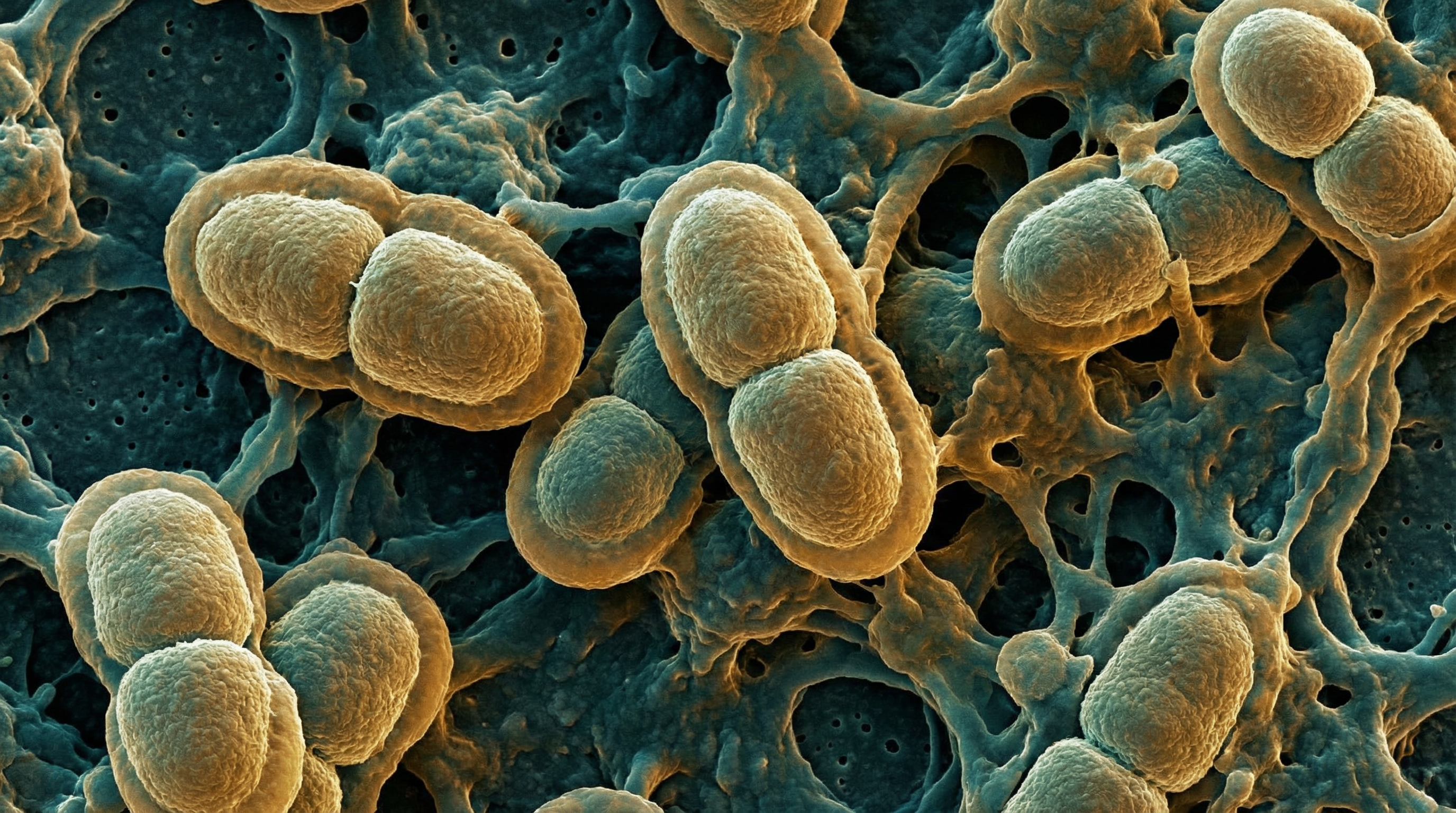
Acinetobacter baumannii is a Gram-negative, aerobic, non-fermenting coccobacillus and one of the most clinically significant opportunistic pathogens in healthcare settings. Designated as a WHO priority pathogen for new antibiotic development, it is a member of the notorious ESKAPE pathogens and causes up to 1.4 million hospital-acquired infections annually.[1]
Key Characteristics
A. baumannii possesses several distinctive features that contribute to its success as a nosocomial pathogen:
- Morphology: Pleomorphic coccobacilli appearing in pairs or chains
- Environmental persistence: Survives for weeks to months on dry hospital surfaces
- Biofilm formation: Forms robust biofilms on medical devices and surfaces
- Genomic plasticity: Contains numerous mobile genetic elements facilitating resistance gene acquisition
- Natural competence: Capable of DNA uptake through horizontal gene transfer
- Metabolic versatility: Utilizes various carbon and energy sources
Antibiotic Resistance Mechanisms
A. baumannii has developed an alarming array of resistance mechanisms making it increasingly difficult to treat:[2]
β-Lactamases
- Produces all four Ambler classes of β-lactamases
- OXA-type carbapenemases (OXA-23, OXA-24, OXA-51, OXA-58) are predominant
- Metallo-β-lactamases (NDM, IMP, VIM) increasingly reported
Efflux Pumps
- AdeABC: Primary efflux pump conferring multidrug resistance
- AdeFGH and AdeIJK: Additional RND-family pumps
- Up-regulation contributes to tigecycline and colistin resistance
Other Mechanisms
- Decreased membrane permeability through porin loss (CarO, OprD)
- Target site modifications (lipid A modifications for colistin resistance)
- Homologous recombination for acquiring resistance traits[3]
Virulence Factors
The pathogen employs a "persist and resist" strategy through diverse virulence factors:[4]
Adhesion and Biofilm
- Outer membrane proteins (OmpA, CarO, Omp33): Facilitate host cell attachment
- CsuA/BABCDE pili: Essential for abiotic surface attachment
- Bap (Biofilm-associated protein): Critical for biofilm maturation
Immune Evasion
- Capsular polysaccharides: Protect against complement and phagocytosis
- LPS modifications: Alter host immune recognition
- Type VI secretion system: Delivers effectors to competing bacteria and host cells
Metal Acquisition
- Acinetobactin: Primary siderophore for iron acquisition
- ZnuABC: Zinc transport system
- Sophisticated systems for manganese acquisition
Clinical Significance
Infections and Mortality
A. baumannii causes severe healthcare-associated infections with mortality rates of 43-84% in ICU settings:
- Ventilator-associated pneumonia (VAP): Most common manifestation
- Bloodstream infections: Often catheter-related with high mortality
- Wound infections: Particularly in trauma and burns
- Meningitis: Following neurosurgical procedures
- Urinary tract infections: Catheter-associated
Risk Factors
- Mechanical ventilation
- Prolonged ICU stay
- Prior antibiotic therapy
- Invasive devices (central lines, urinary catheters)
- Immunosuppression
- Combat-related injuries ("Iraqibacter")
Treatment Options
Current therapeutic strategies for carbapenem-resistant A. baumannii (CRAB) infections are limited:[5]
First-Line Options
- Sulbactam-durlobactam with carbapenem: Preferred therapy for CRAB
- High-dose ampicillin-sulbactam (6-9g daily): Backbone of many regimens
- Polymyxin B: Favored over colistin due to better pharmacokinetics
Alternative Agents
- High-dose tigecycline (100mg q12h): For non-pulmonary infections
- Cefiderocol: Emerging option, especially for MDR strains
- Minocycline (200mg q12h): Alternative for susceptible strains
Emerging Therapies
- Bacteriophage therapy
- Antimicrobial peptides
- CRISPR-Cas systems for sequence-specific targeting
- Artilysins (engineered endolysins)
Gut Colonization and Microbiome Interactions
Recent research has revealed important aspects of A. baumannii gut colonization:[6]
- Ornithine metabolism: Uses ornithine succinyltransferase (AstO) to compete with gut microbiota
- Dietary influence: Dietary ornithine supplementation promotes colonization
- Infant colonization: Significantly higher in formula-fed versus breastfed infants
- Reservoir function: Gut serves as metabolic reservoir for antimicrobial-resistant strains
- Commensal inhibition: Produces acinetobactin to outcompete skin and respiratory commensals
One Health Perspective
A. baumannii is increasingly recognized as a One Health pathogen found in companion animals, livestock, wildlife, food, and aquatic environments. Direct transmission between human and non-human populations has been documented, with resistance increasing in populations with closer human contact.