Overview
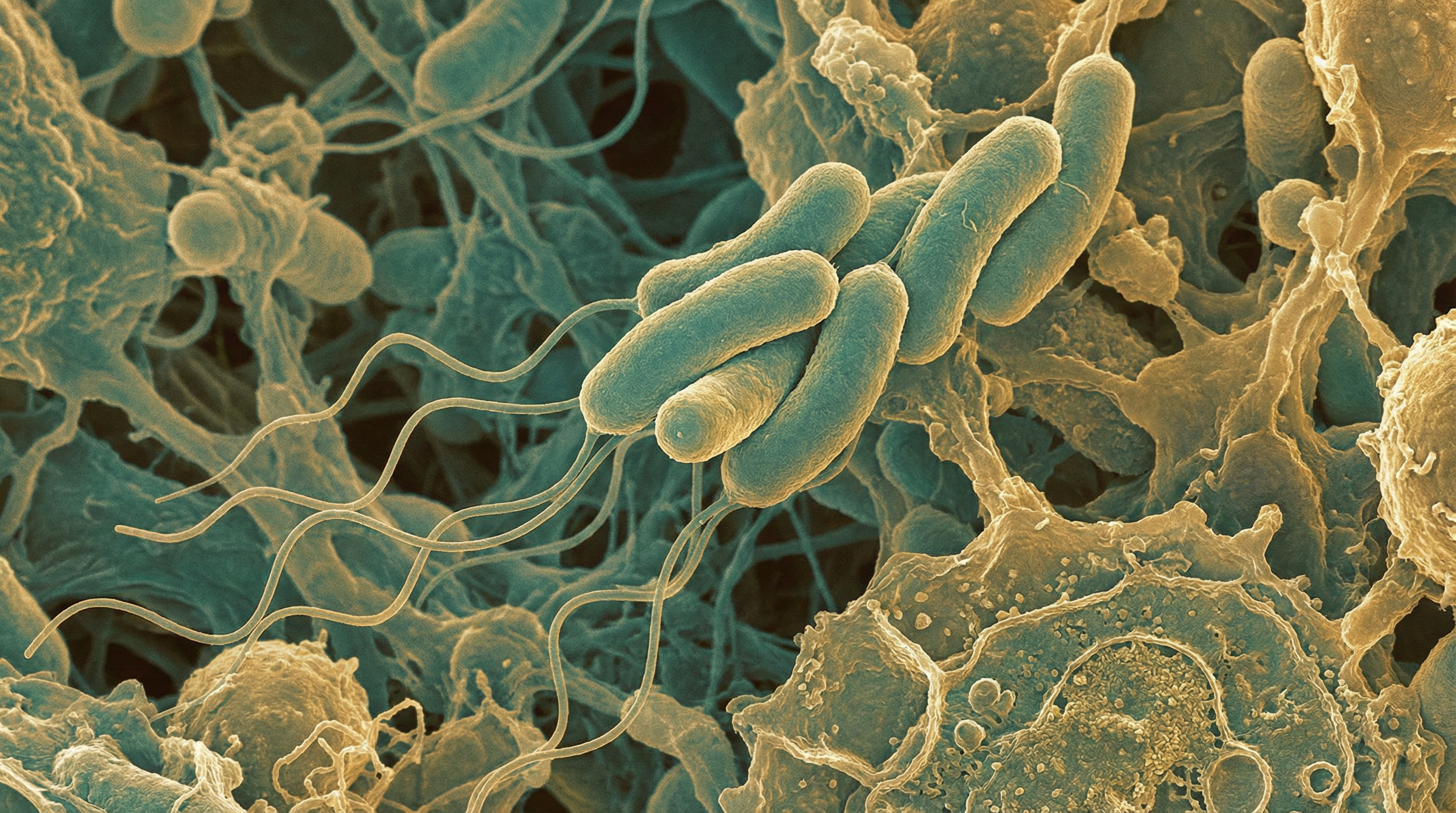
Roseburia intestinalis is an anaerobic, Gram-positive, slightly curved rod-shaped flagellated bacterium that belongs to the phylum Firmicutes, class Clostridia, order Clostridiales, and family Lachnospiraceae.[1] It is one of the most abundant butyrate-producing bacteria in the human gut microbiome, typically accounting for 0.9%–5.0% (mean = 2.3%) of the total microbiota, with Roseburia species collectively representing 7-24% of total bacteria in the healthy human colon.[1] R. intestinalis is increasingly recognized as a key beneficial microorganism with significant potential as a "next-generation probiotic" due to its numerous positive effects on human health.
Characteristics
R. intestinalis is an obligate anaerobe that requires specific growth conditions. It produces butyric acid as a major metabolic end product through the butyryl-CoA:acetate CoA-transferase pathway, with enzyme activity measured at 38.95 ± 3.40 µmol⁻¹ mg protein⁻¹ min⁻¹ during exponential growth phase.[2] The bacterium possesses flagella that allow it to penetrate the colonic mucus layer and interact with the epithelium.
Key features include:
- Morphology: Slightly curved rod-shaped, flagellated bacteria
- Metabolism: Obligate anaerobe that produces butyrate (~2000 µg/mL in culture) as a major end product of fermentation[3]
- Growth Requirements: Anaerobic conditions at 37°C with specific growth rate of 0.44 ± 0.05 h⁻¹
- Genome Features: Contains butyryl-CoA:acetate CoA transferase, the key enzyme for butyrate production; 4,340 coding sequences; 41 genomic islands; 7 prophages[1]
- Substrate Utilization: Can ferment dietary fibers, especially resistant starches, xylan, and β-mannan
β-Mannan Metabolism: A Specialized Capability
R. intestinalis demonstrates remarkable specialization in degrading dietary β-mannans, plant cell wall polysaccharides commonly found in human diets.[4] The bacterium expresses two conserved loci (MULL and MULS) containing specialized enzymatic machinery:
- RiGH26: An extracellular endomannanase that hydrolyzes spruce acetyl-galactoglucomannan (AcGGM) into oligosaccharides within 1 hour
- ABC Transporter System: RiMnBP/RiMPP1/RiMPP2 for internalization (binding affinity Kd = 2.55 µM)
- Decorations Removal: RiCE2 and RiCEX esterases remove acetyl groups; RiGH36 α-galactosidase removes galactose with 100% efficiency
In competitive co-culture experiments, R. intestinalis outcompeted Bacteroides ovatus (72.5% vs 27.5% at stationary phase), demonstrating its role as a primary, specialist degrader.[4] Dietary supplementation with 2.5-7.5% AcGGM produces a 10-30 fold increase in R. intestinalis relative abundance.
Gut Barrier and Epithelial Health
R. intestinalis exerts profound protective effects on gut barrier function through multiple mechanisms:
Tight Junction Enhancement
Studies demonstrate upregulation of critical tight junction proteins including ZO-1, claudin-1, claudin-3, occludin, and TJP1.[5] In 27-OHC-induced cognitive impairment models, R. intestinalis reversed reductions in occludin (F = 24.33, p < 0.0001) and claudin-1 (F = 21.4, p < 0.0001).[5]
Intestinal Permeability
Using FITC-dextran (4 kDa) permeability assays, R. intestinalis significantly reduced intestinal permeability in ApcMin/+ mice and ethanol-challenged Caco-2 cells (p < 0.001).[3][6]
Mucus Layer Protection
R. intestinalis colonizes the cecum and colonic mucus layers, upregulating secreted mucin MUC2 and preventing endotoxin (LPS) translocation.[6]
Anti-Inflammatory and Immunomodulatory Effects
Flagellin-TLR5 Signaling Pathway
R. intestinalis flagellin binds to Toll-like receptor 5 (TLR5) on intestinal epithelial cells, initiating a protective immunomodulatory cascade:[7]
- TSLP Production: Flagellin-TLR5 interaction induces thymic stromal lymphopoietin (TSLP) secretion by IECs
- Dendritic Cell Activation: TSLP activates dendritic cells to secrete IL-10 and TGF-β (>1000-fold increase vs IEC supernatant alone)
- Treg Differentiation: This promotes CD4+CD25+FOXP3+ regulatory T cell differentiation
Importantly, IEC-expressed TLR5—not immune cell TLR5—is critical for these protective effects, as demonstrated by bone marrow transplantation experiments.[7] In Crohn's disease patients (24 CD vs 22 healthy controls), R. intestinalis levels positively correlated with TLR5, TSLP, TGF-β, and IL-10 expression (p<0.01 or p<0.001).
NLRP3 Inflammasome Inhibition
R. intestinalis flagellin upregulates miR-223-3p, which targets the NLRP3 3'-UTR, resulting in:[8]
- Reduced NLRP3 inflammasome activation
- Decreased caspase-1 cleavage
- Inhibition of Gasdermin D-mediated pyroptosis
- Reduced IL-1β and IL-18 secretion
In DSS-induced colitis models, flagellin treatment (50 mg/kg IP daily) significantly restored body weight, decreased disease activity index scores, and inhibited serum IL-1β, IL-18, TNF-α, and IL-6 (p<0.05).[8]
Cancer Prevention and Immunotherapy Enhancement
Colorectal Cancer Protection
R. intestinalis is significantly depleted in CRC patients (444 CRC vs 575 healthy controls across 5 cohorts).[3] The bacterium provides protection through multiple mechanisms:
Butyrate/TLR5/NF-κB Pathway in CD8+ T Cells:
- Butyrate binds TLR5 on CD8+ T cells (Surface Plasmon Resonance affinity: 264 μM)
- Activates NF-κB signaling with nuclear translocation of p65
- Enhances production of granzyme B, IFN-γ, and TNF-α
- TLR5 expression increases >10-fold in R. intestinalis-treated CD8+ T cells
Radiosensitization via OR51E1/RALB Autophagy: R. intestinalis butyrate activates the G-protein-coupled receptor OR51E1, triggering RALB signaling and autophagy-mediated CRC cell death.[9] G2/M arrest increased from 24.9% (control) to 30.88% (radiation) to 37.52% (radiation + butyrate).
Anti-PD-1 Immunotherapy Enhancement
Perhaps most remarkably, R. intestinalis administration significantly improves anti-PD-1 efficacy in microsatellite instability-low (MSI-low) and microsatellite stable (MSS) colorectal tumors—tumors typically resistant to checkpoint immunotherapy.[3] This effect is mediated through enhanced cytotoxic CD8+ T cell infiltration and activation.
Metabolic Health Benefits
Type 2 Diabetes
R. intestinalis serves as a marker for classifying type 2 diabetes, being consistently depleted in T2DM patients across 345 individuals from multiple global cohorts.[1] The bacterium increases IL-22 production, which restores insulin sensitivity and alleviates diabetes.
Liver Disease
In a twin cohort study (410 Korean twins), Roseburia abundance was inversely associated with alcohol consumption (r=-0.0005, p=0.0002).[6] Administration improved survival in chronic alcoholic liver disease from 37.5% to 66.7%. Roseburia abundance also inversely correlates with liver fibrosis and AST levels.
For NAFLD, Ruminococcaceae (including Roseburia) shows inverse correlation with fibrosis in non-obese patients (p=0.0012), with microbiome-metabolite combination models achieving AUC=0.939 for predicting significant fibrosis.[10]
Cognitive Health and Gut-Brain Axis
R. intestinalis demonstrates neuroprotective effects through the gut-brain axis. In 27-OHC-induced cognitive impairment models:[5]
- Novel Object Recognition Index improved (p=0.0132)
- Y Maze Spontaneous Alternation improved (p=0.0011)
- Morris Water Maze platform crossings increased (p=0.0089)
- Restored PSD-95 and BDNF expression
- Normalized brain m6A methylation (F=12.14, p=0.0028)
Safety Profile and Probiotic Potential
Comprehensive safety assessment of R. intestinalis L1-82 demonstrates an excellent safety profile:[1]
| Parameter | Result |
|---|---|
| LD50 | >1.9 × 10⁹ CFU/kg (exceeds highest dose tested) |
| NOAEL | 1.32 × 10⁹ CFU/kg/day for 28 days |
| Cytotoxicity | None observed in NCM460, HT-29, Caco-2 cells |
| Hemolytic activity | Negative |
| Gelatinase activity | Negative |
| Virulence factors | None detected (VFDB screening) |
Antibiotic Susceptibility
R. intestinalis is sensitive to ampicillin, vancomycin, gentamicin, erythromycin, clindamycin, tetracycline, and chloramphenicol, meeting EFSA safety requirements.[1]
Formulation Challenges
As a strictly anaerobic organism, R. intestinalis presents manufacturing challenges:
- Requires oxygen-free cultivation and storage
- Limited shelf-life compared to aerotolerant probiotics
- Must survive gastric transit to reach the colon
Potential solutions include microencapsulation, synbiotic formulations with β-mannan or resistant starch prebiotics, and prebiotic strategies to promote endogenous populations.
Disease Associations Summary
R. intestinalis depletion is consistently observed in:
- Inflammatory bowel disease (Crohn's disease and ulcerative colitis)
- Colorectal cancer
- Type 2 diabetes and metabolic syndrome
- Alcoholic and non-alcoholic liver disease
- Cardiovascular disease and atherosclerosis
- Severe COVID-19 (correlates with elevated inflammatory markers)
- Cognitive impairment
Future Therapeutic Applications
R. intestinalis represents a promising next-generation probiotic with potential applications in:
- Combination with immunotherapy: Enhancing anti-PD-1 efficacy in previously resistant CRC
- Radiosensitization: Improving radiotherapy outcomes while protecting healthy tissue
- Synbiotic formulations: Paired with β-mannan or resistant starch prebiotics
- Precision medicine: Biomarker-guided administration to patients with confirmed depletion
- Cognitive health: Targeting gut-brain axis dysfunction in neurological conditions