Overview
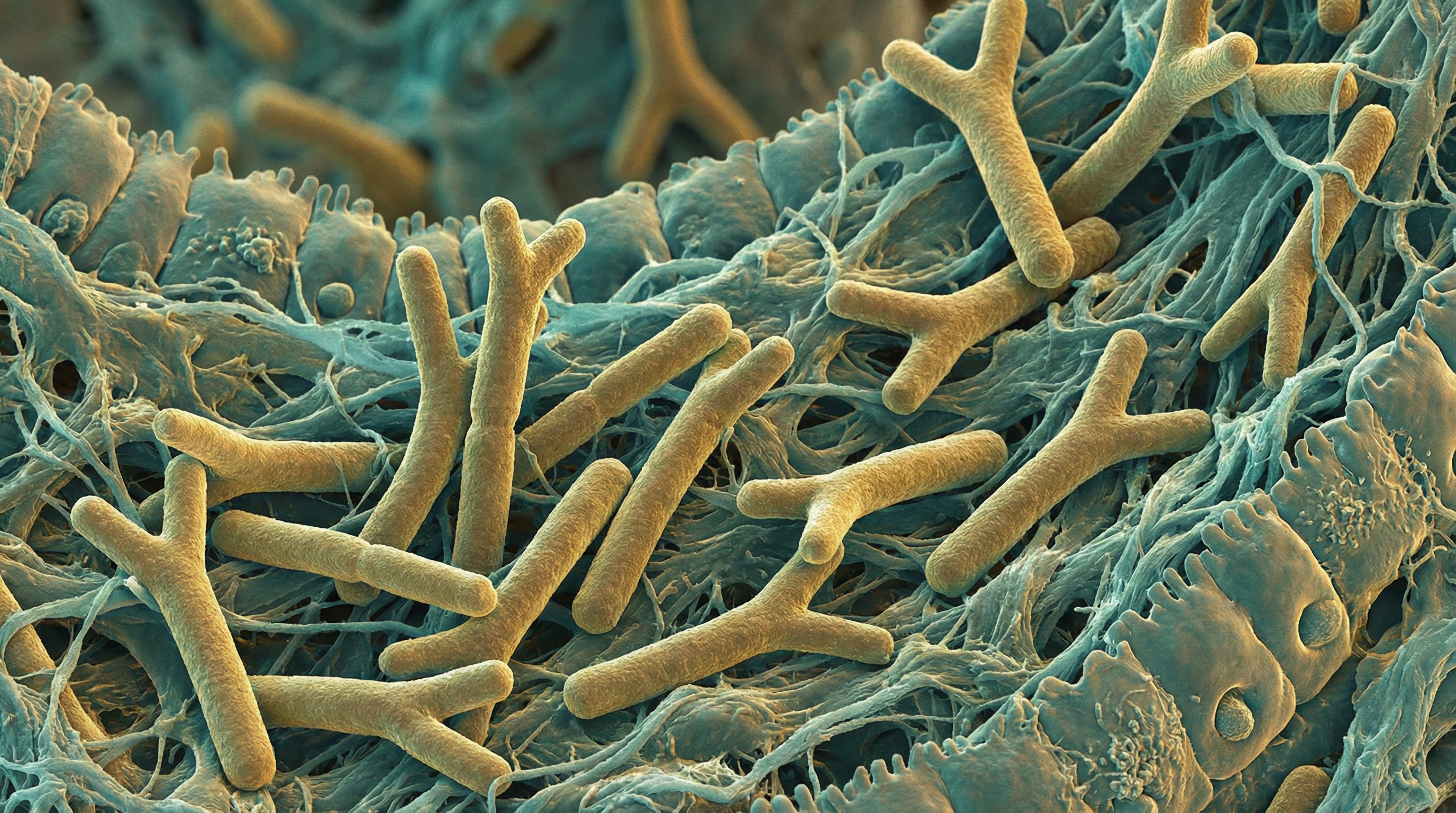
Bifidobacterium longum is a Gram-positive, anaerobic, rod-shaped bacterium and one of the most extensively researched probiotic species. It is among the first colonizers of the infant gut and remains a key beneficial bacterium throughout the lifespan, with documented effects on digestive health, immune function, mental wellbeing, and metabolic health.[1]
Key Strains and Their Applications
BB536
One of the most extensively studied strains, effective for constipation, ulcerative colitis, allergies, and metabolic health.[2]
NCC3001
Psychobiotic strain with documented effects on depression, stress, and the gut-brain axis.[1]
35624 (B. infantis 35624)
Strain with potent immunomodulatory properties, particularly effective for IBS through normalization of IL-10/IL-12 cytokine ratio.[3]
1714
Translational psychobiotic for stress reduction, cognitive enhancement, and sleep quality improvement.[4]
EVC001
Infant-specialized strain that metabolizes human milk oligosaccharides (HMOs) and prevents necrotizing enterocolitis (NEC).
Mechanisms of Action
Immunomodulation
- Upregulation of regulatory T cells (Tregs) expressing Foxp3
- Normalization of IL-10/IL-12 cytokine ratio (anti-inflammatory to pro-inflammatory)
- Reduction of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8, IL-17)
- Modulation of Th1/Th2 and Th17/Treg balance
Gut Barrier Enhancement
- Upregulation of tight junction proteins (ZO-1, occludin, claudin-1)
- Reduction of intestinal permeability
- Activation of TLR2 and NOD2 pathways
- Maintenance of mucus layer integrity
Metabolite Production
- Short-chain fatty acids: Acetate, lactate, and cross-feeding support for butyrate production
- Tryptophan metabolites: Indole-3-lactic acid (ILA), indole-3-carbaldehyde (I3C)
- Biotin synthesis: Produces biotin and pimelate through microbial crosstalk
Gut-Brain Axis Modulation[4]
- Regulation of hypothalamic-pituitary-adrenal (HPA) axis
- Reduction of serum corticosterone and ACTH levels
- Increased Brain-Derived Neurotrophic Factor (BDNF) expression
- Modulation of brain wave activity (theta and alpha bands)
Clinical Evidence
Irritable Bowel Syndrome (IBS)
The strain B. infantis 35624 demonstrated significant reduction in all cardinal IBS symptoms including abdominal pain, bloating, and bowel movement difficulty. The mechanism involves normalization of the IL-10/IL-12 cytokine ratio.[3]
Depression and Anxiety
A landmark 2017 study in Gastroenterology showed that B. longum NCC3001 reduced depression scores in IBS patients with comorbid anxiety/depression. fMRI revealed reduced limbic reactivity to negative emotional stimuli in the amygdala and frontolimbic regions.[1]
Stress Response
B. longum 1714 attenuated cortisol output and subjective anxiety in response to acute stress, while improving hippocampus-dependent visuospatial memory and enhancing frontal midline EEG mobility.[4]
Chronic Constipation
BB536 significantly improved bowel movement frequency and reduced failure of evacuation in elderly individuals with chronic constipation. Mechanisms include SCFA production (acetic and butyric acid) and suppression of microinflammation.[2]
Atopic Dermatitis
B. longum CCFM1029 alleviates atopic dermatitis through conversion of tryptophan to indole-3-carbaldehyde (I3C), activating the aryl hydrocarbon receptor (AhR) signaling pathway and suppressing Th2-type immune responses.[5]
Infant Health
A meta-analysis of 15 RCTs (3,152 infants) found B. longum supplementation significantly reduces risk of necrotizing enterocolitis (RR=0.539). The strain metabolizes HMOs to produce lactic and acetic acids, lowering intestinal pH to inhibit pathogens.[6]
B. longum and B. infantis: Understanding the Relationship
One of the most common points of confusion in probiotic research involves the relationship between Bifidobacterium longum and Bifidobacterium infantis. Taxonomic reclassification has clarified that what was historically called B. infantis is actually a subspecies of B. longum—formally designated Bifidobacterium longum subsp. infantis.
The Three Subspecies of B. longum
B. longum contains three recognized subspecies, each with distinct ecological niches and health applications:
| Subspecies | Primary Niche | Key Specialty |
|---|---|---|
| B. longum subsp. longum | Adult gut | Immune modulation, IBS relief, gut-brain axis |
| B. longum subsp. infantis | Infant gut | HMO metabolism, NEC prevention, immune development |
| B. longum subsp. suis | Animal gut | Less relevant to human health |
B. longum subsp. infantis: The Infant Specialist
B. infantis (now B. longum subsp. infantis) is uniquely adapted to the infant gut environment:
- HMO metabolism: Possesses a complete enzymatic toolkit for metabolizing all human milk oligosaccharide (HMO) structures—a capability that B. longum subsp. longum lacks
- Strain 35624: Originally classified as B. infantis 35624, this strain (now B. longum subsp. longum 35624) is one of the most studied probiotics for IBS, with documented effects on cytokine normalization[7]
- EVC001: A B. infantis strain specifically developed for infant gut colonization; may reduce incidence of necrotizing enterocolitis (NEC) and support immune development[6]
Why This Distinction Matters
Understanding that B. infantis is a subspecies of B. longum has practical implications:
- Product labeling: Some supplements still use the outdated name "B. infantis"—these contain B. longum subsp. infantis
- Strain selection: Despite sharing a species name, B. longum subsp. longum and B. longum subsp. infantis have very different capabilities and optimal applications
- Life stage relevance: B. longum subsp. infantis is most important during infancy, while B. longum subsp. longum (along with B. adolescentis/) dominates the adult Bifidobacterium population
Safety Profile
B. longum has an excellent safety record across all age groups:
- Regulatory status: GRAS (Generally Recognized As Safe) by FDA; QPS (Qualified Presumption of Safety) in Europe
- Adverse events: Rare and typically mild (transient GI symptoms in <5% of users)
- Special populations: Safe in infants, elderly, pregnant women, and immunocompromised individuals
- No antibiotic resistance transfer in safety-tested strains
Dosing Guidelines
| Application | Typical Dose | Duration |
|---|---|---|
| IBS | 1×10⁹ to 1×10¹⁰ CFU daily | 4-8 weeks |
| Constipation | 5×10⁹ to 5×10¹⁰ CFU daily | 4 weeks |
| Stress/Anxiety | 1×10⁹ to 1×10¹⁰ CFU daily | 4-8 weeks |
| Atopic dermatitis | 1×10⁹ CFU daily | 8-12 weeks |
| Ulcerative colitis | 2-3×10¹¹ CFU daily | 8 weeks |
| Infant NEC prevention | 1×10⁷ to 8×10⁹ CFU daily | Variable |