
The phrase "70% of your immune system is in your gut" has become a common refrain in health discussions. While this figure is an approximation, the underlying science is profound: your gut and microbiome together play an essential role in developing, training, and regulating immune function throughout life[1]. The trillions of bacteria, fungi, and other microorganisms residing in the gastrointestinal tract — collectively known as the gut microbiome — act as a frontline regulator of immune health, shaping how the body defends against pathogens, manages inflammation, and maintains tolerance to harmless substances. Understanding this relationship is critical for anyone navigating microbiome-related health conditions.
The Gut: Your Largest Immune Organ
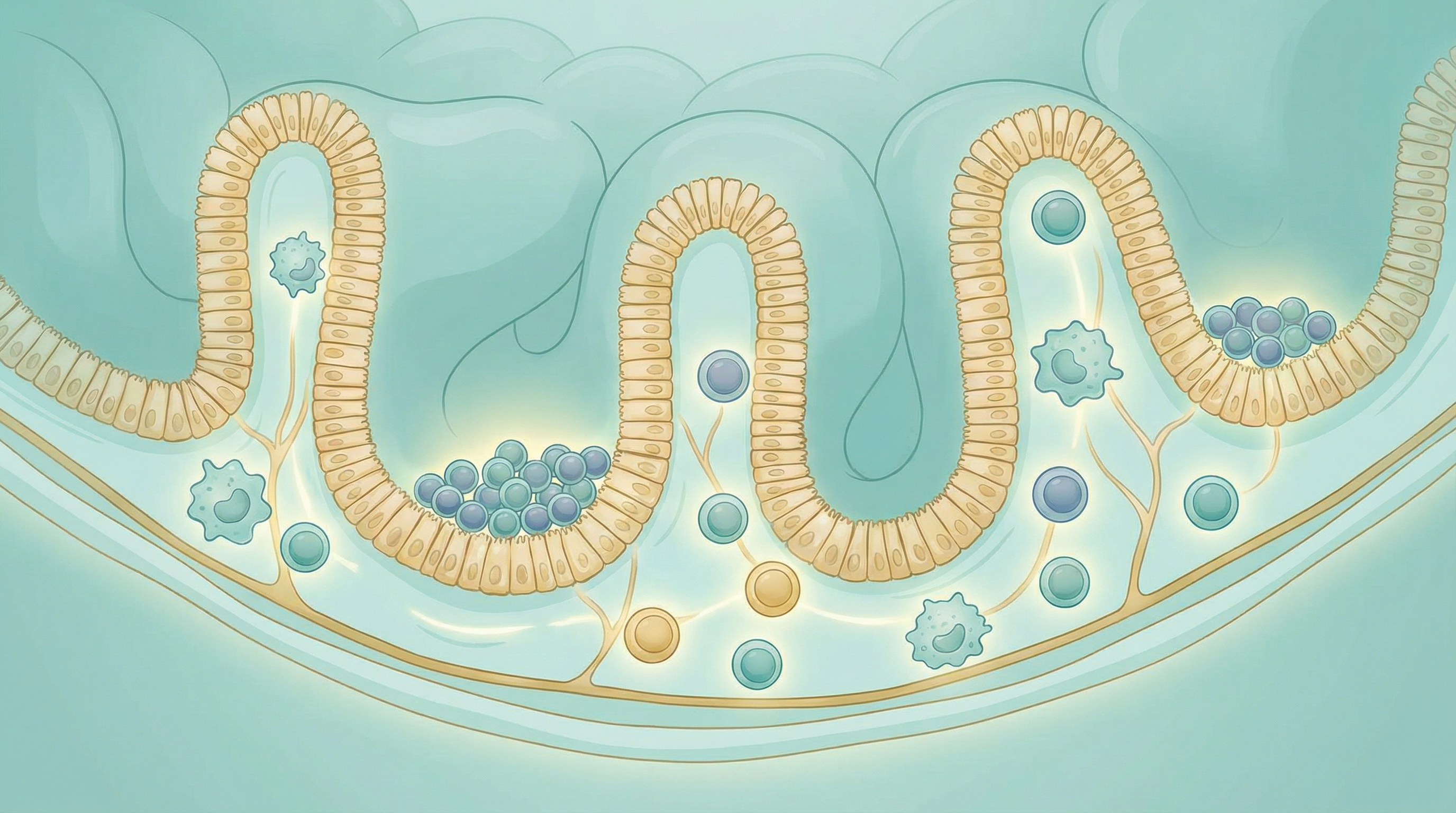
Your gastrointestinal tract houses the gut-associated lymphoid tissue (GALT), the body's largest collection of immune cells. This makes sense from an evolutionary perspective—the gut is the primary interface between your internal environment and the external world of food, microbes, and potential pathogens[2].
Key Immune Structures in the Gut
Peyer's patches: Clusters of lymphoid tissue in the small intestine that sample antigens from the gut lumen and initiate immune responses.
Mesenteric lymph nodes: Process antigens from the gut and generate appropriate immune responses.
Intestinal epithelial cells: Form a barrier while also producing antimicrobial peptides and communicating with immune cells.
Lamina propria: The tissue layer beneath the epithelium, densely populated with immune cells including T cells, B cells, macrophages, and dendritic cells.
How Gut Bacteria Train Your Immune System
From birth, gut microorganisms play a crucial role in educating the immune system[3]. This training process involves:
Early Life Programming
The immune system of newborns is essentially naive. Colonization by gut bacteria during the first years of life is critical for:
- Developing immune tolerance: Learning not to overreact to harmless substances
- Building defense capabilities: Preparing to fight actual pathogens
- Establishing the gut barrier: Creating a functional intestinal lining
Research shows that children born via cesarean section, who miss initial exposure to maternal vaginal and gut bacteria, have higher rates of allergies, asthma, and autoimmune conditions. Similarly, early antibiotic exposure can disrupt this critical training period.
Continuous Immune Education
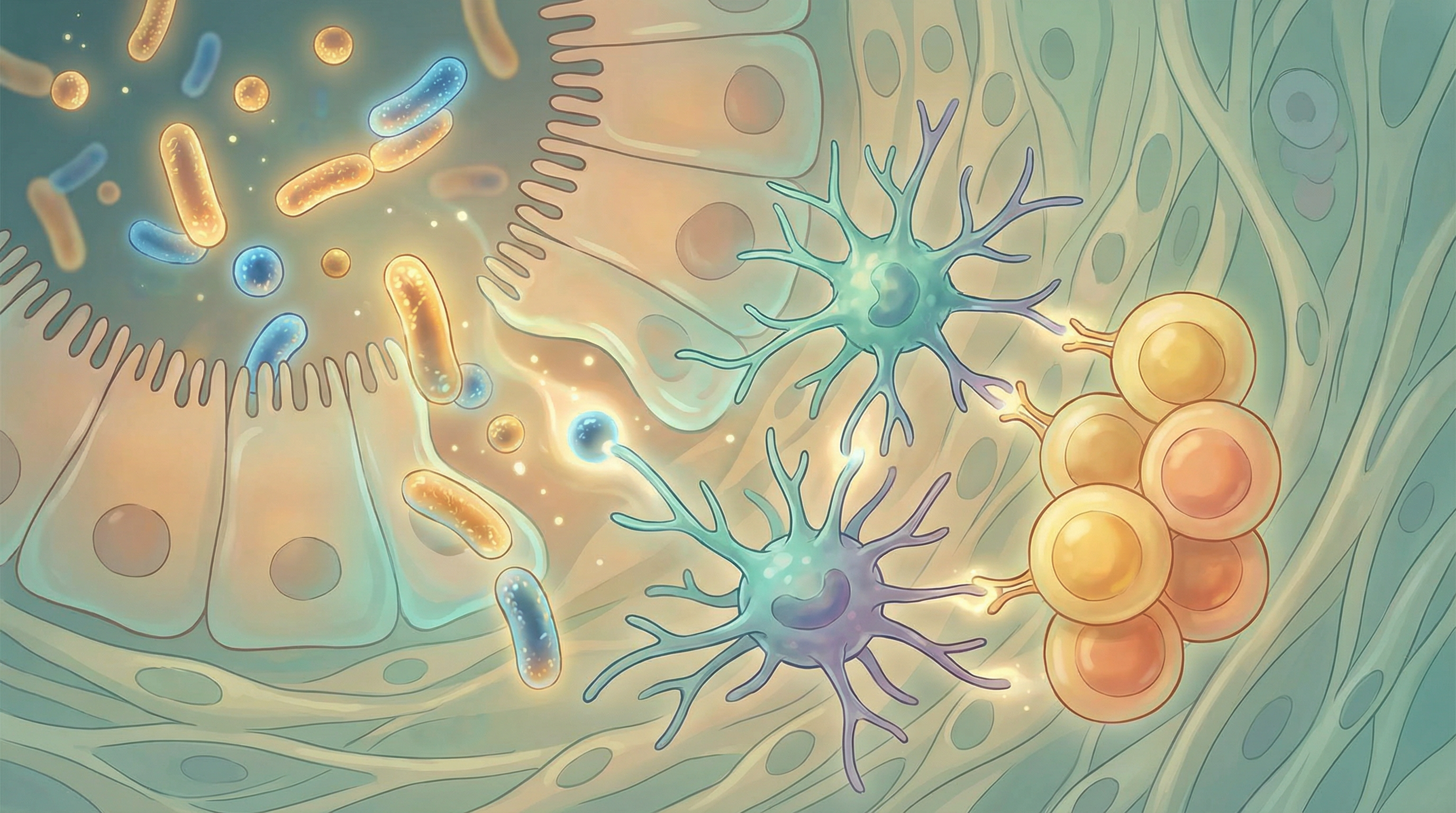
The microbiome continues to shape immunity throughout life. Beneficial bacteria constantly communicate with immune cells through:
- Pattern recognition: Immune cells recognize microbial molecular patterns
- Metabolite signaling: Bacterial products directly influence immune cell behavior
- Direct cell contact: Some bacteria interact directly with immune cells
Key Microbiome-Immune Interactions
Short-Chain Fatty Acids: Immune Modulators
When gut bacteria ferment prebiotic fibers, they produce short-chain fatty acids (SCFAs)—particularly butyrate, propionate, and acetate. These metabolites profoundly influence immune function[4]:
- Promotes development of regulatory T cells (Tregs) that prevent autoimmunity[5]
- Reduces inflammatory signaling in macrophages
- Strengthens the gut barrier, preventing immune activation by gut contents
- Enhances antimicrobial peptide production
- Modulates dendritic cell function
- Influences T helper cell differentiation
- Supports regulatory T cell generation
- Enhances antibody production by B cells
- Supports gut barrier integrity
- Provides energy for peripheral tissues involved in immunity
Specific Bacteria and Immune Regulation
Research has identified particular microorganisms with significant immune-modulating effects[6]:
- One of the most abundant gut bacteria in healthy individuals
- Major butyrate producer
- Has strong anti-inflammatory properties
- Reduced levels associated with IBD and other inflammatory conditions
- Lives in the mucus layer lining the gut
- Strengthens gut barrier function
- Associated with reduced inflammation
- May protect against metabolic disorders
- Produce acetate and lactate
- Enhance antibody responses to pathogens
- Support infant immune development
- May reduce allergic responses
- Modulate dendritic cell function
- Enhance mucosal immunity
- Produce antimicrobial substances
- Support balanced T helper cell responses
Clostridium clusters IV and XIVa:
- Potent inducers of regulatory T cells
- Critical for preventing intestinal inflammation
- Reduced in many autoimmune conditions
The Balance: Immunity Without Inflammation
The immune system faces a constant challenge: it must respond vigorously to dangerous pathogens while tolerating beneficial microbes and harmless food antigens. This balance is maintained through several mechanisms[7]:
Regulatory T Cells (Tregs)
These specialized immune cells act as "peacekeepers," preventing excessive immune responses. Gut bacteria—particularly through SCFA production—are essential for generating adequate Treg populations.
Insufficient Tregs can lead to:
- Inflammatory bowel diseases
- Food allergies and intolerances
- Autoimmune conditions
- Chronic systemic inflammation
The Mucosal Firewall
The gut maintains multiple layers of protection that keep the immune system from overreacting:
- Mucus layer: Physical barrier keeping bacteria away from epithelial cells
- Secretory IgA: Antibodies that neutralize pathogens without triggering inflammation
- Antimicrobial peptides: Proteins that kill harmful bacteria while sparing commensals
- Tight junctions: Seal between epithelial cells, preventing leakage
When the System Breaks Down: Dysbiosis and Disease
Disruption of the gut microbiome—known as dysbiosis—can trigger or worsen immune-related conditions[8]:
Allergies and Atopic Conditions
The "hygiene hypothesis" suggests that reduced microbial exposure contributes to the rising prevalence of allergies. Specific microbiome patterns associated with allergic disease include:
- Reduced diversity in early life
- Lower abundance of Bifidobacterium and certain Clostridia
- Altered SCFA production
- Impaired Treg development
Microbiome-targeted interventions show promise for allergy prevention and treatment, particularly when initiated early in life.
Autoimmune Diseases
Multiple autoimmune conditions show characteristic microbiome alterations:
Inflammatory Bowel Disease (IBD):
- Reduced Faecalibacterium prausnitzii
- Decreased microbial diversity
- Increased potentially pathogenic bacteria
- Impaired butyrate production
- Altered Prevotella levels
- Reduced beneficial Bifidobacterium
- Gut inflammation preceding joint symptoms
Type 1 Diabetes:
- Reduced microbial diversity before disease onset
- Altered SCFA-producing bacteria
- Potential role in triggering autoimmune attack on pancreatic cells
Multiple Sclerosis:
- Distinct microbiome signatures
- Potential gut-brain-immune axis involvement
- Ongoing research into microbiome-based therapies
Chronic Inflammation
Even without specific autoimmune disease, dysbiosis can promote low-grade systemic inflammation—now recognized as a driver of numerous chronic conditions including:
- Cardiovascular disease
- Metabolic syndrome
- Neurodegenerative diseases
- Certain cancers
- Accelerated aging
Supporting Immune Health Through the Microbiome
Understanding gut-immune connections opens powerful opportunities for supporting immune health:
Dietary Strategies
Increase prebiotic fiber intake:
- Inulin from chicory root, garlic, onions
- Beta-glucan from oats, mushrooms
- Resistant starch from cooled potatoes, green bananas
- Pectin from apples, citrus fruits
Consume fermented foods:
- Yogurt with live cultures
- Kefir
- Sauerkraut and kimchi
- Miso and tempeh
Eat polyphenol-rich foods:
- Berries, grapes, cocoa
- Green tea, coffee
- Extra virgin olive oil
- Colorful vegetables
Limit immune-disrupting foods:
- Ultra-processed foods with emulsifiers
- Excessive sugar
- Artificial sweeteners (may alter microbiome)
- Excessive alcohol
Targeted Probiotic Supplementation
For immune support, research suggests particular strains may be beneficial:
- Lactobacillus rhamnosus GG: Reduces infection duration, supports gut barrier
- Bifidobacterium lactis: Enhances immune cell activity
- Lactobacillus acidophilus: Supports mucosal immunity
- Saccharomyces boulardii: Protects against pathogenic bacteria
Always choose probiotics with demonstrated efficacy for your specific goal, and consult healthcare providers for personalized recommendations.
Lifestyle Factors
Manage stress: Chronic stress alters microbiome composition and immune function through the gut-brain axis. Practices like meditation, yoga, and adequate rest support both microbiome and immune health.
Exercise regularly: Moderate exercise increases microbial diversity and supports immune function. Extreme exercise, however, can temporarily suppress immunity.
Prioritize sleep: Disrupted sleep patterns alter the microbiome and impair immune responses. Aim for consistent sleep-wake cycles and 7-9 hours nightly.
Minimize unnecessary antibiotics: While sometimes essential, antibiotics can significantly disrupt the microbiome. When antibiotics are necessary, supporting microbiome recovery afterward is crucial.

Testing and Personalization
Microbiome testing can provide insights into your gut-immune connection:
What testing can reveal:
- Microbial diversity (correlates with immune health)
- Levels of key immune-modulating bacteria
- SCFA production capacity
- Potential dysbiosis patterns
Limitations to understand:
- Testing provides a snapshot, not continuous data
- Normal ranges vary significantly between individuals
- Direct causation is difficult to establish
- Results should be interpreted with professional guidance
The Future of Microbiome-Based Immunotherapy
Exciting research directions include:
- Precision probiotics: Engineered strains designed to modulate specific immune pathways
- Postbiotics: Defined bacterial metabolites used therapeutically
- Fecal microbiota transplantation: For severe dysbiosis and immune conditions
- Microbiome-informed drug development: Medications that work synergistically with gut bacteria
Conclusion
The gut microbiome represents a powerful lever for influencing immune function. From early life development through ongoing immune regulation, the bacteria residing in your digestive system profoundly shape how your body responds to threats while maintaining tolerance to beneficial substances.
By nurturing a diverse, balanced microbiome through diet, lifestyle, and targeted interventions, you can support robust immune function while reducing the risk of allergic and autoimmune conditions.
Whether you're looking to strengthen defenses against infections, manage an existing immune condition, or optimize overall health, attending to your gut microbiome is an evidence-based strategy with far-reaching benefits.
Explore our immunity health goal for more strategies, or learn about specific conditions where microbiome interventions show promise. Browse the full microbiome database to understand the key species involved in immune regulation, or consider microbiome testing to understand your personal gut-immune landscape and develop targeted interventions.
What the Research Shows: Key Clinical Evidence
The connection between gut microbiome and immunity is not theoretical — it is supported by decades of controlled research:
Germ-Free Animal Studies: Mice raised without any gut bacteria have dramatically underdeveloped immune systems, with fewer T cells, reduced antibody production, and impaired ability to fight infections. When gut bacteria are reintroduced, immune function normalizes — demonstrating that the microbiome is essential for immune development[3].
Regulatory T Cell Induction: A landmark 2013 study in Nature showed that specific Clostridium strains from the human gut microbiota could induce regulatory T cells when administered to germ-free mice — directly demonstrating how gut bacteria maintain immune tolerance and prevent autoimmunity[6].
Butyrate and Treg Promotion: A separate 2013 Nature study confirmed that butyrate (the primary SCFA produced by gut bacteria fermenting fiber) is a key signal that drives regulatory T cell differentiation in the colon[5]. This mechanistic link explains why low dietary fiber — and the resulting low butyrate — is associated with higher rates of inflammatory conditions.
Dysbiosis and Inflammatory Disease: Longitudinal studies in humans show that reduced microbiome diversity and lower F. prausnitzii abundance precede flares in inflammatory bowel disease — suggesting the microbiome change drives the immune dysfunction, not just the reverse[8].
Probiotics in Clinical Trials: A systematic review of 63 randomized controlled trials found that probiotic supplementation significantly reduced the duration and severity of upper respiratory tract infections — one of the cleanest demonstrations that gut bacteria directly influence systemic immune responses to pathogens.
These findings collectively support the microbiome as a primary regulator of immune function — not just a bystander — and validate dietary and probiotic interventions as meaningful strategies for immune support.
Frequently Asked Questions
How does the gut microbiome affect the immune system?
The gut microbiome trains and regulates the immune system through three main mechanisms: producing short-chain fatty acids (like butyrate) that promote regulatory T cells and reduce inflammation; direct interaction between gut bacteria and intestinal immune cells including dendritic cells and macrophages; and maintaining the gut barrier to prevent inflammatory bacterial products from entering the bloodstream.
What percentage of the immune system is in the gut?
Approximately 70% of the body's immune cells reside in and around the gut, within the gut-associated lymphoid tissue (GALT). This includes Peyer's patches, mesenteric lymph nodes, and the lamina propria, making the gut the largest immune organ in the body.
Which gut bacteria are most important for immune health?
Key immune-supporting bacteria include Faecalibacterium prausnitzii (anti-inflammatory butyrate producer), Akkermansia muciniphila (gut barrier integrity), Bifidobacterium species (antibody responses), Clostridium clusters IV and XIVa (regulatory T cell induction), and Lactobacillus species (mucosal immunity).
Can improving gut bacteria help with autoimmune disease?
Research suggests yes, though clinical application is still evolving. Patients with IBD, rheumatoid arthritis, multiple sclerosis, and type 1 diabetes consistently show altered microbiome signatures. Prebiotic fiber, specific probiotic strains, and dietary pattern changes have shown benefit in reducing inflammatory markers in clinical trials. Consult a healthcare provider for personalized guidance.
What diet best supports gut immune health?
A diverse, plant-rich diet high in prebiotic fiber (inulin, beta-glucan, resistant starch), fermented foods (yogurt, kefir, kimchi), and polyphenols (berries, green tea, olive oil) consistently shows the strongest evidence for supporting gut immune function. The Mediterranean diet is the most well-studied pattern in clinical trials for this purpose.
Frequently Asked Questions
How does the gut microbiome affect the immune system?
The gut microbiome trains and regulates the immune system through three main mechanisms: producing short-chain fatty acids (like butyrate) that promote regulatory T cells and reduce inflammation; direct interaction between gut bacteria and intestinal immune cells including dendritic cells and macrophages; and maintaining the gut barrier to prevent inflammatory bacterial products from entering the bloodstream.
What percentage of the immune system is in the gut?
Approximately 70% of the body's immune cells reside in and around the gut, within the gut-associated lymphoid tissue (GALT). This includes Peyer's patches, mesenteric lymph nodes, and the lamina propria. This makes the gut the largest immune organ in the body.
Which gut bacteria are most important for immune health?
Key immune-supporting bacteria include Faecalibacterium prausnitzii (anti-inflammatory butyrate producer), Akkermansia muciniphila (gut barrier integrity), Bifidobacterium species (antibody responses, early immune development), Clostridium clusters IV and XIVa (regulatory T cell induction), and Lactobacillus species (mucosal immunity, antimicrobial peptides).
Can improving gut bacteria help with autoimmune disease?
Research suggests yes, though this is still an active area of study. Patients with IBD, rheumatoid arthritis, multiple sclerosis, and type 1 diabetes consistently show altered microbiome signatures. Interventions including specific probiotic strains, prebiotic fiber, and dietary patterns have shown some benefit in reducing inflammation markers. Clinical application is growing but should be guided by a healthcare provider.
What diet best supports gut immune health?
A diverse, plant-rich diet high in prebiotic fiber (inulin, beta-glucan, resistant starch), fermented foods (yogurt, kefir, kimchi), and polyphenols (berries, green tea, olive oil) consistently shows the strongest evidence for supporting gut immune function. The Mediterranean diet pattern is the most well-studied in clinical trials.
References
- Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121-141. doi:10.1016/j.cell.2014.03.011
- Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Research. 2020;30(6):492-506. doi:10.1038/s41422-020-0332-7
- Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature. 2016;535(7610):75-84. doi:10.1038/nature18848
- Rooks MG, Garrett WS. Gut microbiota, metabolites and host immunity. Nature Reviews Immunology. 2016;16(6):341-352. doi:10.1038/nri.2016.42
- Arpaia N, Campbell C, Fan X, et al.. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. 2013;504(7480):451-455. doi:10.1038/nature12726
- Atarashi K, Tanoue T, Oshima K, et al.. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature. 2013;500(7461):232-236. doi:10.1038/nature12331
- Blander JM, Longman RS, Iliev ID, et al.. Regulation of inflammation by microbiota interactions with the host. Nature Immunology. 2017;18(8):851-860. doi:10.1038/ni.3780
- Levy M, Kolodziejczyk AA, Thaiss CA, Elinav E. Dysbiosis and the immune system. Nature Reviews Immunology. 2017;17(4):219-232. doi:10.1038/nri.2017.7